|
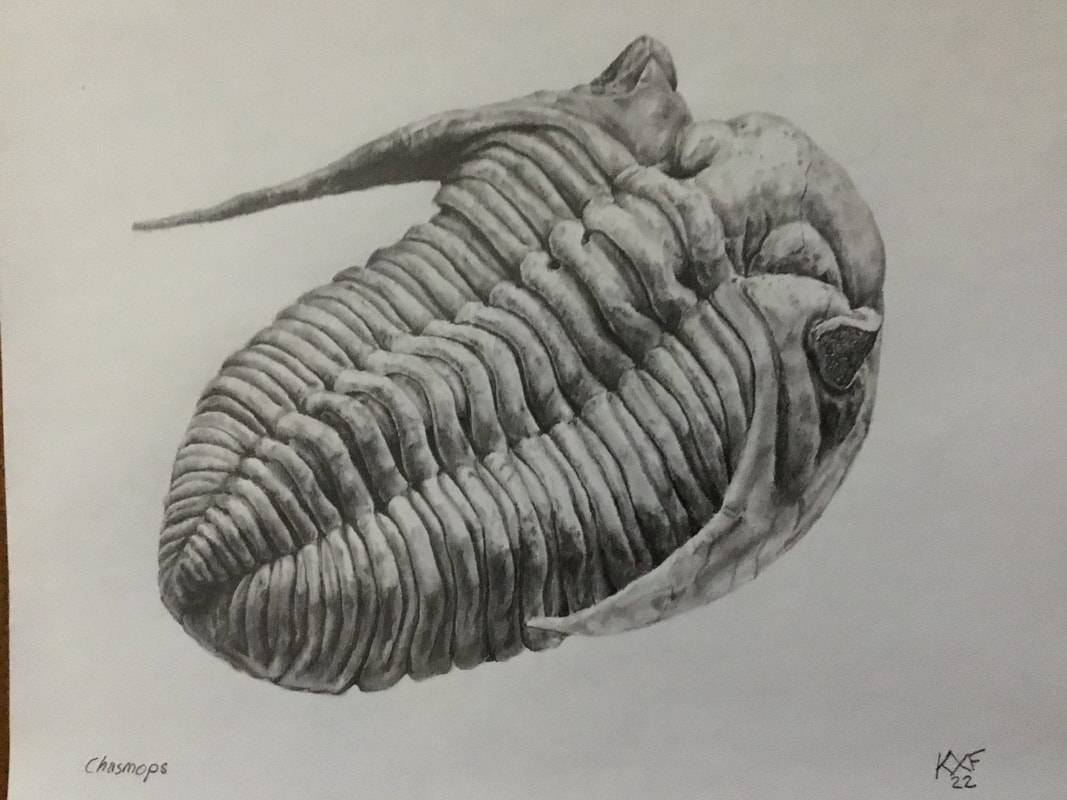
The warmer weather is back, but sadly not well timed since I will be back teaching and grading through the last 5ish weeks of the semester. That said, I had some time off this week to look more closely at my finds from the Ordovician out east, and found two surprises. The first and lesser surprise was this. I had picked up this thin plate on account of a scattering of 3-4 tubercular bumps with an intention to see what it was all about. You never know: it might be something like an echinoderm. After a bit of abrasion, I could rule out starfish and claim this is a fragment of Bufoceraurus, bringing me to 15 encountered species of trilobite so far in 2024. The bigger surprise is much smaller (as in, a few millimetres). I need to trot out the better camera as the iPad zoom leaves something to be desired, but this is the cranidium of a very rare cheirurid, Kawina trentonensis, which is only known by... its cranidia. I've added the image from Hessin's book below my specimen to show how it favourably compares. So, not only the 16th trilobite encountered in the field for 2024, but also my first new species of the year.
It's great to get a running start on the fossil season, although it remains to be seen if my luck holds. Historically, the season starts out strong, falls into a ditch by summer, and then picks up a bit in the autumn. Pending the weather in March, I may go on a quick day trip on the train to the lower mid-Devonian to find a few more of the usual suspects in Onandaga material, and then in mid-April I have designs on some other Ordovician spots. Then, it's teach a week in May and maybe back on a train for more fossil fun. It is passing rare up here when there is no snow and the temperatures are warm enough to get out in the field. Typically at this time of year, the ground is covered in a thick white blanket that won't be pulled back until late March or early April, but the El Niño effect has kept our winter well above seasonal, but that is due to flip shorty as Winter is Coming (back), and likely with a walloping of snow. The last two weekends have seen me digging locally, and at a spot east of Toronto. The local Devonian material was not all that generous, and the Ordovician material out east just marginally more so despite all the expense and time spent. Nevertheless, any fieldwork in February is a win onto itself. I'll start with the non-trilobitic items first, or at least the ones I bothered to take home. The Ordovician material (Bobcaygeon and Verulam Formations) was nowhere near as productive as my site visits in the past. Just about all the usual trilobites were present, but nearly all strictly as fragmentary. On the upper left is a crinoid (I have not checked the genus and species in the literature yet), and it looks "murky" as I had to slather it in glue to prevent it flaking off. To the right is a Pleurocystites that may or may not have all its plates, but does seem to have both anterior projections with one of them buried. Most likely a Carabocrinus judging by the shape. Not a prize specimen, but it fell loose from a slab. This is likely Ceraurus plattinensis that was perched atop a rubbly layer of Chondrites. It came right off the slab, and it is uncertain if the pygidium is there or tucked somewhere. In the image on the upper left, a glance to the upper right shows another cheirurid partially buried. I did indeed collect the complete right genal spine that I will be gluing back on, and possibly gluing the entire specimen back in place on the slab. The bottom row shows the specimen in both dorsal and ventral aspects, with the ventral side clearly showing the hypostome. Another double in the lumpy-bumpy Chondrites layer, more cheirurids, and I'll need to look closer to assign these as Gabriceraurus dentatus, Ceraurus globulobatis, or a mix of both. The devil is in the ornamentation details, as well as cephalic widths. The uppermost specimen is in pretty rough shape, while the bottom one may be buried enough to be salvaged. I will still look to preparing them both as a double on a plate is nice. This cheirurid was the second chum on the same plate as the C. plattinensis that kicked things off. It may be buried or weathered away -- only prep will say. On the right is a complete prone Flexicalymene senaria, which are not terribly exciting, but it rests in a slightly tilted fashion (a common orientation in this material for the prones of the species) on a very thin flake.
I have some other pieces I took home for closer examination, but they are entirely speculative at this point. Many other fossils were encountered and left in the field on account of my not needing heavy nautiloids or endless bryozoans and brachiopods. It was not very generous or forgiving material, either; some heartbreakers occurred during attempts to extract them without power tools. Saturday was a balmy, sunny day, whereas Sunday saw a precipitous drop to a cloudy just-a-notch-over-freezing day. Still, I can at least say I got out and did it. Does this count as the season starter? Not quite. This was anomalous, and I only count a season opener when there is going to be consistency in the weather that has winter firmly behind it. Although my heart is in fossils as I await spring and the new fieldwork season, my mind is occupied by the semester (and my body daily committed to the gym). There have been no notable fossil-related events for some while now, but I did manage to squeeze out some time to get into the box on the weekend to prep out one of my October finds from Hastings County. As found on the left, and some preliminary scribe work on the right before I set it aside. It is passing rare that these appear fully robust as opposed to crushed (as this specimen is, sadly), but it is mostly complete and represents the only full prone of this species that I have found. I am tentatively labeling it Bumastoides bellevillensis, but am not entirely certain on the species designation here. And in its completed state. Despite this overhead shot, it is quite plump and 3D, which is nice. Due to the head being somewhat bashed in, I opted not to risk digging too deep around the cephalon for fear of risking damage.
I have a number of other specimens that deserve my attention, although my abrasion unit is working as well as a centenarian blowing out birthday candles. At present, I also have tinkered a bit with a giant semi-prone calymenid that will also need some restoration. With money being a bit tight (and a need to save up for some trips this year), I have not gone hunting in the shopping formation for any new trilobites in 2024 yet, so I will likely put the buying on ice while I focus on the prep surplus and focus more on field collecting to supply my growing collection of trilobites. Never one to pass up punishment, I made the decision to expand my planned book on Devonian trilobites of southwestern Ontario to include all trilobites of Ontario. Many of us look fondly upon Ludvigsen's famous green trilo-bible published by the ROM in 1979, but much has happened since and it is my Sisyphean task to revise it with the current knowledge, to put another brick atop the masterful foundation and legacy Rolf Ludvigsen left us.
At present, much of the Devonian is complete and stands at 90(!) pages, including lithological descriptions and, of course, featuring the trilobites. I am chasing down photos from premium collectors in Ontario with full attribution. If you are an Ontario collector with complete specimens or even fragments of rare trilobites, do not hesitate to reach out. A work like this is a legacy project, and possibly a capstone to my days in the field. I cannot give a forecast as to its completion, but I will be working steadily on this over the winter. The book will NOT be erecting new species or any other such shenanigans. This will be a workmanlike field guide aggregating our knowledge of Ontario trilobites. It will be a big lift, but fortunately works by William Hessin and Philip Isotalo among others will help move the marker forward, as those are field guide bibles in their own right. It will take a village, so to speak. My plan is to make the book freely available to everyone. I seek no profit from these labours, and consider this my thank you to every intrepid collector and researcher in the field. As stated, if you have premium specimens from Ontario that should be featured, I am but an email away. Just as the sound of the bell signals the closing of the market day, so too does a more figurative bell signal the time to down tools and take stock in the collecting season that was. And now here I am, putting my tools and kit away in the spare room so that they no longer clutter the dining room to my wife’s frustration — well, I’m getting around to it at any rate! It is a time to put away the tools and rocks in their proper places and brace for winter’s frosty hold. It is of course customary to start the planning process all over again for the next season, dreaming big with all that open time and possibility ahead of us, that blank canvass of the calendar for what may come when next we raise hammers again. And, indeed, winter is a time to organize and curate the finds, to prepare those specimens, to draw, to deep dive into research with a goal of directing the next season’s tireless hunt. Yet, the season’s closing is first and foremost a time of reflection, casting the memory backward into the season that was. The season for me never lives up to what I had hoped for, yet it always seems to yield up so much more than I could ever have expected. This year was no different. Much blood and treasure went into this season, and like the Rolling Stones lyric goes, “you can’t always get what you want, but you get what you need.” So many failures, disappointments and misadventures dotted the season and yet — and yet! — it has been a spectacularly memorable season. I added 22 new species of trilobite to the collection, 13 of which were new field finds (the rest purchased or gifted). I encountered 45 distinct species of trilobite spanning over 40 days in the field, matching if not exceeding my incredible 2020 season when we were under Covid lockdown. I acquired new specimens most collectors may never have an opportunity to possess. I did not, however, succeed in finding anything new to science, which remains a shining goal. I traveled the equivalent of a quarter of the earth’s circumference. I went deep and far, tight and local, and got to delight in spending time with so many incredible field comrades, too. I am saddened that I have to be unbearably circumspect about where I went; it wasn’t always like this. Now it is time to sum up the season in my usual way, showing the best finds, preps, purchases, and even doodles (which I need to go back to in a serious way this winter). TRIPsI can be pardoned for many omissions in this public report, although everything is logged in my field notebook. My season began on March 26th when I ventured to a local ditch by a new housing development. Oddly enough, the first trilobite encountered in the field that day was Ordovician on account of some big retaining wall blocks imported from the Gull River Fm, and so pieces of Isotelus were scattered here and there in mostly the gastropod-littered rock with its deep forest green glauconite mineral replacement. By early April, my semester was winding down and I made some further local visits, and one ride-along with a friend to inspect a gravel pit that turned out to be mostly Devonian trash salad. And yet we did find a few old slabs of Craigleith shale at an abandoned property that we took back to my place and split. The first Devonian bugs were all moulted fragments of the usual proetids around these parts, but some nice Pseudogygites hash plates were pretty fun. A few more local site visits until I boarded the first of many trains, circa mid-April, to a Devonian spot about an hour east, which saw me encounter parts of three more trilobites, bringing my season tally to a dozen. I bumped into a piece of my old friend Terataspis again by close to April’s end, in a local stormwater management pond, but it would not be long after that when I would have to temporarily swap the tools for the lectern for my standard week-long grad course on strategic planning. Train trip #2 would be a whiplash tour through Ottawa and eventually Quebec City. I would leave the very next day my class ended to meet up that evening with an old poet friend’s son who had asked me to read and critique a draft of his novel. A fun night was had at my favourite punk rock tavern, but I had to be up early the next morning to see about a site on the far west of the city. The site turned out to be lacklustre with very scrappy material. On the following day, I made my way to a shale dump that was surprisingly rich in pyritized Triarthrus rougensis and ridiculous loads of nautiloids and graptolite beds. I even found some pyritized sponges which are destined to be donated to a museum in Montreal. And then it was all aboard the next train to Quebec City to collect with my dear fossil comrade. At this point, I had added five more trilobite encounters to the season’s list, bringing me up to 18. About four distinct locations were hunted in the Quebec City area, including an area replete with occasional beds of white mineral replaced Cryptolithus moults. The usual suspects of Ceraurus pleurexanthemus and Flexicalymene senaria were found, but nothing I would qualify as A-grade. We also discovered a new spot that had parts of Meadowtownella sp., but they were sparse in very ugly material. There was still ample material collected thus far, which meant I had to leave some surplus with my friend to mail back to my house — keeping in mind I was living entirely out of my already heavy field pack and duffel bag. As there were no trains going all the way west from Quebec City to London in the same day, I booked an overnighter motel and returned to my shale dump for one last poke before boarding the homebound train. All told, it was 11 days away from home and I missed wifey and kitten, and desperately needed a shave. June was a relatively quiet month. I did board the train a third time, but for work purposes in running a facilitation session for Brampton council. Zero collecting to be had around there. We had already planned to do a short visit to see Deb’s auntie up around Barrie/Collingwood way, and that gave me the opportunity to do a 3/4 day poking around a road cut. Unlike the last time I went there in the autumn on the way back from Temiskaming, the only complete trilobite found was a Flexi roller. The material was Cobourg Fm equivalent, but not the more stunning examples as may be found in the south. Still, three more trilobite species encountered to bring the season tally to 24. The remaining days of the month saw me do some local site visits, and eventually board the train for the fourth time back for another day trip to my eastern Devonian prospect. I then did one recon prospect that turned out to be nothing. July is usually the lull in my season, so apart from two local stops, most of it was dedicated to logistics planning for an August adventure. And what an interesting August trip that was that saw me away from home for 20 days. Train ride #5 saw me go to Quebec City again and then off on perhaps one of the most memorable adventures filled with every conceivable bit of bad luck one can imagine, yet we earned our stripes and still came back with goodies. It could be said that this was the most productive trip for trilobite encounters, adding nine more to the season tally, eight of which were new to my collection. Of course, I came back feeling a bit broken, and 20 days felt like 200. It was good to be home. By month’s end and the beginning of the fall semester nigh, I was visited by two US fossil comrades. We had plenty of laughs and fun, and they wanted to see the famous Arkona. On the following day, we did the longer drive to Formosa Reef where we found the usual fauna, and I was able to add two more trilobite species encounters to bring season tally to 35. By September, teaching was in full swing, so not a lot of hammer work was done. I did one desultory tour of local spots, finding just one sad fragment of a proetid. Usually at this time of year the weather is still a bit too hot, but also infested with yellow jacket wasps recently kicked out of their hives and acting crazy. Near month’s end, I did one other local stormwater pond spot and found the usual Bois Blanc Fm examples, including a rather sad piece of Terataspis. October is generally one of the best months for collecting. Weather has cooled down a bit, and the flora has thinned out a bit more to make for easier seeing and navigating. Given where we park Thanksgiving so early up here in Canada, that effectively granted me a four day weekend to board the train a sixth time to venture east to a spot in the Ordovician of Ontario. I had visited this site back in late May 2022. I was not expecting great riches, but then was completely surprised with all the finds that added six trilobite encounters, two of which were new to the collection (Raymondites longispinus, “Xylabion” Ceraurinella sp.), bringing my season tally to 41. Closer to month’s end, a few local spots that yielded much of the same. And then with Reading Week, I took the opportunity to board a train a seventh time to return to my eastern Ontario spot only to get partially skunked despite that Hibbertia find, and then back out to Quebec City to see my friend a third time this year. Sadly, winter had been an early visitor, which curtailed our collecting to a single half day. I returned home only partially victorious after a week’s absence, my pack a bit lighter than I would have liked. That brings me to November when I did do a local ditch spot for not much of any gain. I then did a day outing with my dear fossil friend Matt to some Devonian spots outside of the city to close out the season. [more here] It is always a bittersweet occasion to finally put the tools to bed, but eventually one runs out of runway — or, more specifically, warm days in which to get out in the field without it being very uncomfortable. My usual cut-off is around 5 celsius for a day high; anything lower than that results in cold hands (even in work gloves) and impaired vision trying to strain to see in the colder air. Much less daylight hours and the imminent threat of snow means each day is a throw of the dice, and eventually there are more losers than winners as far as collecting days are concerned. I ended the season today, November 19th. We did the very last place on our list in the Bois Blanc Fm in search of Terataspis and we got skunked. As disappointing as that may sound, I am no longer left wondering and we did our due diligence. Steadily over three years we crossed sites off the list. is the hunt over? No way. This is when we start getting creative and expansive in our search, embracing the prospector’s way of life in more adventurous ways. Bittersweet as ending the season with a whimper and not a bang may be, at least now we know, and it completed our long list. In all, the 2023 season ran 238 days, from March 26th to November 19th, out of which I saw 43 days of field action. This has been a very good and interesting year. My regrets are minor when stacked against the experiences had. FINDS & AcquisitionsThe lucky 13 added to the list are not all complete, of course, but no less welcome to the growing collection. It wasn’t until August that I bagged the first new find for the collection. I didn’t take out the credit card so much on purchases this year as I seemed to have spent more on travel for field collecting. Still, I grabbed a few, and was gifted a few others, giving me 9. Here is a list of all 22 trilobites added to the collection, either by field or purchase or gift: Thysanopeltis sp. Hoekaspis yahuari Damesella paronai Ciliscutellum ciliensis Anacheirurus sp. Kettneraspis sp. (Bolivia) Ceraurinus icarus Acernaspis orestes Diacalymene schucherti Stenopareia grandis Arctinurus anticostiensis Failleana wangi Illaenus bayfieldi Isotelus harrisi Raymondites longispinus “Xylabion” (Ceraurinella n.sp.) Hibbertia ottawaensis Bumastoides bellevillensis Triarthrus sp. Redlichia sp. Wanneria sp. Synphoria stemmata Only five of the trilobite species new to my field collecting were complete. I did have some luck finding complete specimens of trilobites that were already logged from previous years, so it was a good haul. A selection of a few new acquisitions here: PREpSI had a lot of bad luck at the bench this year when two of my only scribes crapped out. I was able to purchase two others, which did save the day. My abrasive unit is still crappy and temperamental until I can upgrade to a Swam, COMCO, or equivalent. What started with not much great material to work on in the winter then eased into not being at the bench very much due to active collecting. There are a number of preps on the go, yet not complete, but eventually I'll post them over the next while. DoodlesI simply did not put enough time at the table this year, and yet I have a long queue of bugs to draw. I hope to nurture those ambitions in our colder months when work allows. ResearchI do likely have some new material for research, but the nub of it is that there are just not that many researchers in this niche part of the world. In trilobites of Ontario and Quebec, we have longstanding housekeeping issues that need to be worked out, but that is a question of bodies, interest, and funding. How long will we be stuck with Isotelus “mafritzae” or assuming our Ceraurus pleurexanthemus is identical to that found in Quebec and Rust Walcott when they are clearly different? How about cleaning up our cheirurids, or revisiting our long neglected Devonian trilobites of Ontario? These things may never happen in my lifetime, but I’m not going to be so presumptuous and vain to try to erect new species on my own either. I’ve seen how that goes when overly ambitious amateurs start splitting genera into a dizzying number of “new” species, and it means by ICZN rules that those names get removed from use in the future. Many times, species splitting is just a marketing gimmick to sell the same species twice under different names to satisfy the completist. In terms of my own engagement with research, I continue to be fed by a great trilobite expert, so I lack for nothing in that regard for my reading pleasure on the trains (which I take a lot of!). With some of my close trilobite workers, I have waded into debates where it seems lives hang in the balance, such is the passion among this tiny 100ish global cohort. There is also always the usual drama and scandals, natural to such a small group. Next stepsI am already beginning a preview and planning for 2024. There remain longstanding commitments to sites I keep rolling over each year, but there are no shortage of long distance prospects for the coming season. With work out east in mid-April, I may as well fold that into a longer adventure. There are some hazy plans to do work with a rostroconch expert, and a host of farflung locations that would just make sense to visit. There may also be some periods of prospecting, and that is usually a low percentage success rate as far as these things go, yet worth the risk if the reward is commensurate. Ending off here with whatever other random items appear on this year's photo fossil roll. Enjoy! I got my dates all mixed up thinking Reading Week was the following week, but I was wrong, which meant that I had to hustle to book train and motel, toss everything into the trusty field pack and bag, and boogie. It was time to get out into the field for the work-free week. The journey would see me heading out east again into the fickle riches of the Ordovician of Ontario, and eventually Quebec. Setting the tone of the adventure was during my train layover in Toronto when a not-so-sane person wanted to pick a fight with me, which I would say is a fairly unwise life decision, particularly looking as I do wearing steel-toed boots with a bag of hammers. But my keen diplomacy de-escalated the situation and I was off on the rails to my first destination. Sadly, Saturday saw my initial site inaccessible, and I made the 25km trek on foot to another location that turned out to be a nothingburger. I retreated to my motel room and returned to my initial spot the next day. This was the spot that had yielded so much glory in my October visit, but had decided to be fickle and parsimonious this time around. Here are two scrappy Flexicalymene senaria. Sadly, the same cheirurids that were quite profuse in the last visit had gone MIA. A complete enrolled Bumastoides I extracted from a bloc. As was par for my luck on this trip, this specimen is now missing. Having checked all the gazillion pockets in my field pack and clothing, it is still MIA. I am hoping it will magically turn up at some point. It is just too cute to lose forever! This was by far the trip-maker. Although the yield was depressingly low for a week-long trip, this find on Day 2 is quite special, and had been sitting out weathering for years! Unlikely to be complete, but this is a ventral of Hibbertia ottawaensis, the rarest harpid we have in Ontario. A handful of specimens are known. Upon the recommendation of a friend, I have reached out to a professional preparator to perform a transplant and flip rather than make the attempt on my own and risk botching such a scarce example. Updates on this one to follow, no doubt. On the Monday, I took the next train to Quebec City to visit my friend who had located a new river valley source with some peculiar Triarthrus not typical of the region. When I arrived, so did winter. The site is highly contingent on there being no snow, so we had to wait it out for a few days, chewing the fat and drinking some beer.
The Triarthrus are indeed unique in their way. Although we failed to find any complete, there are at least two different species there. The nodes along the axis of a few of these suggest T. beckii. But note the size and 3D aspect to the glabella on some of these samples I took back. My friend suspects these may be a new species, but it's hard to say. The last big revision of this genus were done by Ludvigsen and Tuffnell a long time ago. So that was my trip: a lot longer for a lot less, but weather and circumstances intervened. The harpid was the highlight, ticking off a new species in my Ontario list (now up to 69). It won't be long until I write up my 2023 season wrap-up, so stay tuned. I may get out a few more times, but I will likely be keeping things more local. This past trip seems to capture the motif of my 2023 season, though. Misadventures and dashed expectations, yet the occasional "wow" find despite it all, making this year a sort of "good, despite the conditions and circumstances." A Blend of Romanticism and Reality I am occasionally asked what it means when I say, “I’m off on a dig for a few weeks.” Whatever do I get up to in the field? For those who have never experienced the thrills and challenges, I can park a few words here. One of the immense pleasures of fossils is in the nature of conducting work in the field, a composite of hair-raising adventures mixed with the tedious grind, the excitements and utter disappointments, a full spectrum of emotions that rarely are situated in the mushy mediocre middle. Generally, these events cleave to the absolute highs or lows. It involves a lot of logistical planning, pre-trip research of poring over maps and old literature, and being in nature on its terms. There have been times of fear and joy as we go about this trade in a spirit of liquor and guessing. The past year has been no exception in this regard. I’ve covered over 10,000 km by road, rail, and boot. I have trekked far into the hinterlands, over rugged terrain, through thick tangles of the bush, navigating pristine waterways, swatting away bugs, sweating or freezing pending the time of year, contending with high tides and rock falls. I have also been in urban settings where fossils sit in an island of civilization, untroubled and ignored by the quick bustle around them. Some sites are a breeze, and others are a treacherous slog into the unknown. Not every field trip is a pioneer adventure into the wilds. Some of them are quite safe and banal. I wouldn’t want to give the impression that every time I stepped out for a dig I am wrestling bears or wading through tabanid-infested muskeg. And there is no doubt that when we fossil comrades get together, we drink heavily! This is something we share in common with our disciplinary cousins, geologists. It is customary that a long day in the field out in the boonies busting rocks until we can’t lift our arms any more is punctuated at the end with just enough strength in reserve to set up camp and tip the elbow. Even that takes planning. Those who must travel several miles up a mountain with a field pack full of tools are not likely to strap on a few two-fours of beer as opposed to the more portable liquor, but where possible a few cases of beer are on hand. It doesn’t matter if the beer is warm. And, no, we don’t drink ourselves stupid since we have to get up early the next day for another round in the field (and then another beery round at the end of the day). Rinse and repeat. To do this well and to score incredible finds, it takes a mix of good planning, rugged determination, fortitude, and physical fitness. Unlike, say, going to an already established fossil park, there is no safety net or easy retreat to civilization. You (and maybe a comrade) are on your own, and that includes taking into account all the contingencies of getting lost or injured. Real fieldwork is not for duffers. More’s the pity that the trend in earth sciences education has been to minimize fieldwork requirements in favour of pulling out map drawers to study specimens in the lab. Sure, that has its vital place in the increase of our knowledge, but who will fill the drawers? When I read the literature of those fieldworkers of old, one can almost feel that bracing adventure out in the wilderness, investigating untouched strata and collecting specimens in all sorts of weather. One becomes accustomed to carrying one’s bodyweight in tools and rocks on one’s back for many, many miles. At the end of any expedition, you and your vehicle should be equally dirty and broken. If going solo (not always recommended, but sometimes necessary), it can get a bit lonely, and the snatches of the stupidest songs may get caught in a loop in your head, try as you might to tune it out and listen to the sound of leaves or babbling brooks and bird calls and all that. Some people enjoy rucking and hiking for its health benefits. In our case, we get the exercise incidentally through the work towards a goal. One has to account for every pound you take into the bush, and every pound you take out. Carrying tools is one thing, but also adequate water, which can be drank or dumped to make room for finds on the way back from the dig. I know every time I go on one of these weeks-long adventures, I tend to be 5-10lbs lighter because of all the exertion and forgetting to eat. A long field expedition is a great way to get that beach body, albeit filled with cuts, bruises, and the appearance of a shaggy, dirty hobo. Failure is always possible. Vehicle failures in the middle of nowhere? Been there. Tool failure? Yup. Site failure in not yielding up anything to make the trip of any value? More times than I can count. I would say my batting average is just under half the trips I go on result in some measure of success, be that incredible or so-so. Good planning helps mitigate against that kind of failure, but even all the best planning in the world cannot prevent brute reality from tossing failure grenades in your path. Failure is the most probable in prospecting new sites, but it is in this act of pioneering and discovery that presents the highest risk/reward ratio, just as it was during the gold rush. I have gone prospecting and came away with riches or empty-handed, with the latter being more the case. It can be treacherous. It is not just having to deal with nature, but with other collectors who may be keeping an eye on where you are going, ready to look for any signs of location to steal a march on your parade. For that reason, being incredibly circumspect or even silent is the most prudent option. A competitor will deplete your honey hole faster than you may think, no matter how remote, as anyone motivated enough will try to seize the hoard. Posting here on the internet is a surefire way of putting a big sign on your special spot, and before you know it word gets around and it gets raided by many people. It does not matter that you did all your due diligence in establishing that site through hours or even days and weeks of satellite mapping and LIDAR, scratching around in the dirt: giving that information away means you’ve done all that work for other people who can just saunter in and take it. The Gear Most of us would feel naked without our hammers, and we can be real snobs about them. You also contend with the mystery of why you have so many intact left-handed field gloves and hardly any right-handed ones. In the run-up to a trip, you may be raiding liquor stores for both drinkable provisions and flats for specimen storage, and making a trip to other stores to get tin foil and superglue. The chisels, the hammers, the specimen wrapping materials, the glues, the beer — all tools of the trade. At times, a rock saw, but that depends on site (no sense carting a rock saw and gas for a 10 mile bush hike). Fieldwork is no fashion show, yet we do have our ‘uniforms” of sorts that are entirely practical. The ability to bundle up or strip down (or having a change of clothes if one gets soaked) is key. Blasting out the knees and ass of pants is common. Old, sturdy clothes with lots of pockets is beneficial. I usually will wear army pants and sometimes don a surveyor’s vest. On my head a trusty hat, and on my feet a rugged steel-toed clodhopper boot. Expect that clothes will get sweat-stained and ripped. When out in the bush, shelter is a necessity. Over time getting used to another man’s stink, particularly if you share a few weeks in a 2-man tent is all part of the adventure. I find expeditions so much more meaningful with a field comrade: someone to share beer with, to bleed with, to delight in discovery, to commiserate over disappointments, to plan next steps, to meet challenges together, and those rolling conversations late into those very dark nights. The Elements There will always be those things outside of one’s control, and although one can prepare for just about any contingency short of asteroid strikes or sudden wombat attacks, one must contend with the fickle nature of weather. I have planned for months to go on a trip only to get rained out for days even in the statistically driest month of the year. It is beyond pointless to look at weather forecasts so far out as it is to predict the stock price of a company on any given day. The farther out the forecast, the less likely it is to be accurate anyhow. At best, you take it as it comes and prepare accordingly. Perhaps on the day prior to travel, looking at the radar (not the forecast!) is far more informative and predictive. Sticky, Blaring-hot sunny days mean keeping hydrated, cool, and preventing sunburns. Rainy, wet days are just miserable overall and may limit what one can see in the field. Snow is a nuisance that buries rock under an unhelpful additional layer of strata, not to mention if it is cold enough to freeze the ground so that even picking up a piece of talus requires chiseling it out of the frost. Hurricane force winds and thunderstorms sort of put a damper on collecting specimens. It is not just the meteorological phenomena we have to contend with, but also its aftereffects, such as flash floods that swell rivers or turn an otherwise dry area into sucking mud. If temperatures plunge to below freezing, it is all well and good to bundle up (and add more weight), but it may still be a bit uncomfortable, particularly on even gloved hands (doing delicate hammering work with thick gloves is about as good as that sounds). In the cold, what would be a minor ouch from hitting a rock or missed tool strike is amplified considerably. Death and Safety
The more remote the work, the higher the probability of sudden or creeping death. None of us set out with death in mind beyond unearthing long dead things. I have been to areas where cell signal coverage is nonexistent, and the nearest trace of civilization would take a day of trudging through the bush. I find it always pays to tell key people where I am going in the event that I go missing and my carcass may not be found for months, years, or ever. There is also the chance around tall cliffs for a fatal rock fall, and not even a hardhat is going to be much of life-saver from a car-sized rock falling from 30 feet. There is also a chance that wildlife will not take kindly to one’s benign trespass. Or you may lose your food and water, now having to rely on nature itself. No matter how well you prepare, shit happens, and inasmuch as you can pack for every contingency, there is a balance to be struck as well: how much of that can you realistically cart into the bush? The operative thing is to remain cool and collected in a crisis, managing one problem at a time, but also avoiding unnecessary risks. Sure, you may need to ford a raging river using ropes and risk being washed away, or maybe it means taking the extra time to go upstream to a calmer part of the river even if it adds a few more hours of travel time cutting into collecting time. There are those eventualities that one can never account for, such as being alone and having a heart attack, but that is the risk. In the end, prospecting and fieldwork is both an art and science, but on the whole it also remains liquor and guessing. Every one of us who have traveled deep and far into the field has campfire stories to tell, and although I have several, I hope to continue adding to them for as long as this body allows. Every time I venture out I say goodbye to my wife and cat, not knowing if I will return. The song that haunts me on the way out: www.youtube.com/watch?v=CqO4vBSLHMw (Alice in Chains, "Don't Follow:") As I'm in between trips, here is a short spacer on consolidants, which I use both in the field and in the lab. The arsenal is tiny, but mighty and versatile. I'm not including any of the other materials generally used for restoration, such as Bondo or Milliput, but just the plastics I need in both the field and lab. Superglue (cyanoacrylate): This is, bar none, the most essential of consolidants. Not having enough of this is sometimes the difference between going home from a trip a winner or living in an ocean of regret. This fast-bonding agent is cheap and a must-have in my field pack. These sell at hardware stores, but I don't like buying crazy glue at premium prices, and so opt instead for getting these one-use containers at a dollar store. It is effectively liquid plastic that hardens in air. The other benefit of one-use tubes is there is no fussing around trying to reopen them ever again -- I find the bigger tubes available at hardware stores harden near the cap no matter how airtight you screw it back on, rendering the rest of the tube useless. In the lab, it also has its uses. I've used it to glue two or more halves of rock together for preparation where a trilobite comes out in pieces. The one drawback is in its leaving of a kind of whitish crust, and it is not always easy to abrade off (forget trying to scribe it off as it is like removing caulking with a screwdriver). The trick with superglue is less is more. Putting too much superglue when combining two pieces together can leave a gap, a bit like the filling of an Oreo cookie. Also, if applying directly on a fossil surface, it can be a bit of a nightmare to remove the excess. In that case, one trick to thin it out is to blow on it to make it spread more thinly. I use a thin cyanoacrylate. There are more viscous varieties, but I generally want to abide by the less is more principle and make any joins seamless. When stabilizing a fossil in the field, always pay attention to where the rock is cracking, and wick those with glue; if a crack is visible on the edge of the slab, chances are it is running all the way through the whole rock including the fossil! When dealing with flaky shales, it is important to wick glue in all the little crevices, too, to avoid it flaking off and ruining a great fossil in transit. One of the great things about superglue is that it is "reversible" by dissolving in acetone. It isn't going to be magic, though, so don't expect a few drops of acetone to miraculously remove all the glue you accidentally gooped all over your fossil. At best, it will weaken the bond a bit, allowing you to *gently* pick it off with a sewing needle or similar. Try never to dissolve the whole rock in a big bucket of acetone if you've joined pieces together because they will fall apart. Paraloid: This one is new to my consolidation toolkit. It functions similarly to superglue, except these come in already hardened beads that are dissolved in acetone. Pending how viscous and strong you want it (allowing or not allowing "give" in the bond) will depend on the ratios. I usually go for anywhere between 5-20% paraloid to acetone. I put these in a small glass jar, pour in the acetone, seal and wait. I'm not sure how long this can be stored, but it has to be sealed as air will harden the whole mess. I find it dries fairly quickly, and I can strengthen the bond by applying numerous coats. A good idea is to make more than you anticipate needing. A bit of a challenge to tell in this photo with the glare, but this is the underside (ventral) of a trilobite I want to prepare dorsally. That said, it is pretty much wafer thin (a good portion of the dorsal side is already exposed). There is no way to do prep on this without good, solid consolidation. I'm effectively plasticizing a base, and have applied numerous coats of paraloid.
In the case of the specimen above, both superglue and paraloid served their purposes. In the field, the specimen was showing in a very large rock with a whole lot of flakiness and cross-bedding, so I needed to stabilize that with superglue, including any and all exposed bits of shell prior to strategically removing it with hammer and chisel with very precise, measured strike at very tactical angles with a good understanding of where the rock's structural weaknesses were. There was really no way this was coming out in less than two pieces, but the extraction was a success. As the shell is very brittle, too much force in extraction would have resulted in shattered bits that would have been lost forever. Once I got home, I superglued the two pieces together after ensuring no debris between the two faces to be joined remained. Once set and cured, I then applied the coats of paraloid. None of what I am writing here is new to any person who has spent time fossil digging or preparing. Nevertheless, glue is what truly binds in increasing the chances of having a great versus botched find. It is that time of year when the fossil weather is not too hot nor too cold. I took the opportunity to forsake Thanksgiving festivities to go out in the field, bothering the Bobcaygeon Fm. It started out with a bit of a bang. A nice cluster plate of Ceraurus globulobatis and at least one ventral Gabriceraurus dentatus. A lot of pieces need to be reattached, and there is the possibility that more may be lurking under the matrix. I found this within a half hour, so that somewhat set the tone for the weekend. There is one very nice specimen in the mix, and the rest we'll just have to see what prep shows. A severely exfoliated Calyptaulax callicephalus and the pygidial spines of a cheirurid (likely Ceraurinus marginatus). Although I did encounter a few Calyptaulax on this trip, none of them were really worth bringing home. Heartbreaker ventral Gabriceraurus of healthy size. Despite much care (and glue), this one did not survive extraction. On the right is mostly the impression of a Failleana indeterminata; I did not find even a whisper of this styginid beyond this. The phantoms of Ceraurus, and what I suspect is a Ceraurinella ("Xylabion") on the right. Just impressions, sadly. Either the pygidium is gone, or likely tucked flat sandwich-style underneath on the left Ceraurus, and on the right an enrolled example. It isn't the Ordovician of Ontario without some Flexicalymene senaria. Scrappy examples, maybe worth a quick clean. Pygidium of Ectenaspis homanolotoides, which are fairly rare asaphids. On the right is one of the better examples of Raymondites I found. It is tough to believe, but for my many weeks in the Ordovician of Ontario, I had never managed to find a complete Bumastoides milleri. Sure, plenty of parts, and even complete thoracopygons, but this trip finally crossed that off the list. Photo on the right is just a quick ten minute scribing to expose a bit more before the official prep can reveal this one in its more robust glory. The cephalon is a bit crushed, so not a top shelf bug. A Carabocrinus vincourtlandti complete calyx, and possibly a smaller example on the upper left. Echinoderms are not really my thing, but I will pick them up if they are interesting. Prep will be needed to see if the stout arms are on these. The classic Pleurocystites squamosa. A scrappy example, but the nicer ones refused safe extraction, so this was my consolation prize. The wispy tail is present, but mostly buried under the matrix. A ventral asaphid of about 11-12 cm. Obviously, extraction is going to be tricky business. I used up the last of my glue to stabilize it as best as possible before delicately and slowly using hammer and chisel at key points in the rock to minimize damage. The long diagonal crack shown here runs very deep, so there is no choice but to take it out (carefully!) in two pieces to be glued back together later. Much glue is used to prevent pieces of shell from flying off. Success. Ventral and dorsal views. This is going to be one heck of a prep job requiring much paraloid to stabilize the ventral area. Those pleurae are hanging out matrix-free in the open. I also kept the negative which has the left eye. Although there is not yet agreement between me and my fossil comrade, I am thinking it is an Ectenaspis. It seems a bit too elongated to be a typical Isotelus gigas (looks more like Isotelus "mafritze", but those do not occur in the Bobcaygeon). The one way to determine this beyond a shadow of a doubt is to see if there is that classic projection on the pygidium, which is still mostly buried here. If so, then this is truly the trip-maker.
I have a few other specimens that need to be pieced back together, and photographed. In all, not a bad trip. Despite the surplus of trilobites here, the material is fickle in both preservation and abundance, so requires a lot of time scanning slowly with care. I do hope to get out one more time in early November, assuming weather and opportunity cooperates (coinciding with Reading Week, as otherwise I'm doing the teaching thing). As the weather cools, some of these specimens will be making their rounds in my prep area, so at least I have a few more winter projects to share. Things have been extra busy as of late from the work perspective, but I have managed to scratch the fossil itch a bit. I am hopeful of getting out and about this weekend, and have already booked a three day trip over the Thanksgiving weekend, after which I can post some finds. For now, a quickie. I acquired some corynexochida for the collection. What looks like a series of Matryoshka dolls is five Ciliscutellum ciliensis from the Hunan province, China, Silurian in age. These are typically found in nodules, and WYSIWYG. Although nowhere near perfect, a decent representation of the species from a location I am not likely ever going to see in my lifetime. I've wanted a Thysanopeltis for a while, and the price was truly a steal for this one. Again, not the very best, but a good representative. Not much difference between this and the typical/common Devonian scutellids that come out of Morocco except the fringe of pygidial spines. It is unclear what purpose these served, be it some minor form of defense or courtship. In any event, this would make corynexochid species #28 in the collection, accounting now for more than 10% of the 234 distinct species I have cluttering my small home. The buyer I acquired this specimen and the others above has made my saved top sellers list, and I am likely to acquire more from him on account of some very unique offerings at very reasonable prices.
I suppose it is now one foot into the weekend, a possible prospecting mission on the immediate horizon, and the long weekend trip to look forward to. My schedule may be clogged with work obligations, but the season is not quite yet done with me yet. Now is not yet the time to take stock in all the travels and adventures of 2023, but to keep pushing forward until the tank is empty. |
Kane Faucher
Archives
February 2024
|
































































 RSS Feed
RSS Feed